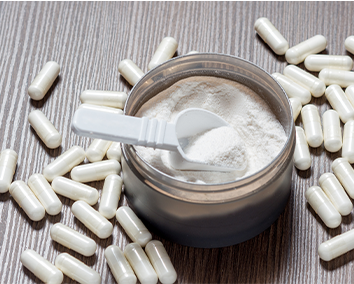
THIRD-PARTY BANNED SUBSTANCE CERTIFICATION PROGRAM COMPARISON
KEY ELEMENTS OF THIRD-PARTY CERTIFICATION FOR BANNED SUBSTANCES
Third party certification for banned substances is vital for athletes, military service members, and first responders when considering dietary supplements or other consumer products as the presence of hidden drugs could lead to a positive drugs test and destroyed career. To understand how the protection in banned substance certification applies to you it is important to evaluate important key elements. Differences in testing frequency, testing menu, reporting levels and process, and transparency in program details and database listings can lead to significant differences in the protection each program offers. We explain what to look for in third-party certification for banned substances in sport in the comparison chart and discussion below, which reviews BSCG® Certified Drug Free vs NSF Certified for Sport® vs Informed Sport / Informed Choice vs HASTA™ vs Cologne List®. A pdf is available through the following link: Comparison Chart – Third Party Banned Substances Certification Programs
Comparison Chart of Key Elements in Third-Party Certification for Banned Substances
BSCG® Certified Drug Free vs NSF Certified for Sport® vs Informed Sport / Informed Choice vs HASTA™ (Human and Supplement Testing Australia) vs Cologne List® (Kolner List)
WHAT IS IMPORTANT TO CONSIDER IN BANNED SUBSTANCE CERTIFICATION?
The list of banned substances is vital. If this is not disclosed you should ask why not. In general, the more drugs included in the testing the broader the protection. BSCG covers more than 507+ drugs, almost double the protection in other programs. A great way to evaluate the relevance of a testing menu from an anti-doping perspective is by looking at the percentage of World Anti-Doping Agency (WADA) adverse analytical findings or positive drug tests, covered by the menu.The BSCG testing menu covers more than 96%, the best protection available in the industry. The BSCG testing menu is also the only one to focus on drugs not banned in sport with additional protection against more than 209 drugs like pain killers, opioids, muscle relaxers, PDE5 inhibitors, and other drugs that the FDA has found when testing for contaminated supplements adding health and regulatory protection.
BE INTERNATIONALLY COMPLIANT
Testing should meet internationally accepted quality standards for analytical testing laboratories. Testing should be performed in International Organization for Standardization (ISO 17025) accredited laboratories using methods that are included in the scope of accreditation and have been validated for dietary supplements and related products. This ensures the analytical testing meets international standards and has been validated to perform at levels required to protect against inadvertent doping.
BATCH TESTING IS VITAL
Understanding batch testing frequency and verifying that a batch has been certified is key. Frequency of testing is one of the most important components of third party certification for banned substances as it tells you if the protection applies to the individual lot in your hand. Make sure to ask for the sampling scheme used in any third party certification program. Always verify the lot number stamped on your package has been certified by looking it up in the program database. Not all programs test every lot.
DETECTION LEVELS
Detection levels should generally be in the low parts per billion range (ng/g) to be capable of finding trace amounts of substances that could lead to positive drug tests for athletes or other drug-tested professionals. Maximum allowable level per serving thresholds for performance enhancing drugs are inherently risky and should only be used in certain circumstances where scientifically justified.
A strong initial audit process is the foundation good third-party dietary supplement certification. That process should include a 21 CFR 111 Current Good Manufacturing Practice (GMP) compliance assessment or consideration of other regional or product specific requirements that considers supplier qualification process, raw material and finished product quality control, adverse event reporting, recall procedures and other vital elements of the quality control and manufacturing process.
TOXICOLOGY TESTS PROTECT
A toxicology and compliance assessment of ingredients should be the starting point of any third-party certification to ensure certified items don’t contain any banned substances, use legal and legitimate ingredients, and don’t exceed generally acceptable levels for certain substances like caffeine.
Transparency is the cornerstone to third-party certification. Key elements should be clearly outlined so athletes, military service members, first responders, and other consumers know what they are getting. Hidden details, like undisclosed frequency of testing, can create holes that banned substances can slip through. Transparency builds trust.
No program is a 100% guarantee that a product is free of banned substances. Evaluating the key elements outlined here can help you determine the level of protection offered in each program. The comparison chart here on third-party certification for banned substances is updated regularly.
We welcome any questions or feedback!