NAD+: Rules, Risks, and Testing in Athletics and Armed Forces
Mar 16, 2026
NAD+ has transitioned from limited discussions in biohacking forums into more prominent consideration within athletic and military contexts. While the compound is marketed for cellular energy and recovery, its regulatory and safety profile is complex. Some NAD+ products are sold as legitimate dietary supplements while other forms originate from loosely regulated research chemical suppliers that may not adhere to GMP regulations or proper quality control. Differences in form and sourcing presents substantive concerns regarding product quality, possible contamination, and compliance with anti-doping standards or regulations regarding structure function claims. Stakeholders, including athletes, military service members, and product brands, require an understanding of how sport governing bodies and military organizations regulate NAD+ in anti-doping efforts, current capabilities of testing, and the critical importance of supply chain transparency and third-party certification and testing.
What Is NAD+
NAD+ (nicotinamide adenine dinucleotide) is an endogenous coenzyme found in all living cells, where it supports cellular metabolism, energy production, and DNA repair processes. NAD+ is the oxidised form that carries a positive charge (so it may be called NAD+) due to the arrangement of bonds on one of the nitrogen atoms, the non oxidised form NAD, sometimes called NADH, has accepted an electron and a hydrogen atom. NAD+ is often sold in capsules or powders as a dietary supplement, and as such it is a legal ingredient according the the definition outlined in the Dietary Supplement Health and Education Act (DSHEA). Two popular precursors also found in supplements are nicotinamide riboside (NR) and nicotinomide mononucleotide (NMN). NAD+ has also become popular in other forms including nasal sprays, injectable products, or for IV infusion that are often sold alongside research chemicals or unapproved peptides that are not allowed for human consumption. The nasal spray, injectable and IV infusion forms present legal concerns and compliance challenges and in sport drug testing they may not be acceptable. Depending on what type of NAD+ product that is purchased consumers may get a legal dietary supplement or compounded product, while injectable NAD+ products sold as research chemicals often lack systematic quality control, third-party testing, or consistent regulatory oversight.
Oral NAD+ Compared to Injectable or IV Infusion
NAD+ is a relatively large molecule with a molar mass of 663.4 g/mol that is difficult for the gut to absorb. Injectable forms or nasal sprays are different forms that offer improved bioavailability when compared to oral forms. The improved bioavailability is attractive to some users and doctors but they often come with increased costs. The oral dietary supplement forms are allowed to make structure function claims, while the injected and nasal spray forms are not allowed to make such claims. This creates somewhat of a legal grey area as the nasal sprays, injectable, and IV therapies are often sold with anti-aging and other structure function claims attached that are not allowed for non-FDA approved substances. The oral forms are convenient to purchase and use, while the injectable or IV infusion forms require doctors prescription and a medical environment for legitimate use.
The Biological Role of This Coenzyme
NAD+ acts as a molecular carrier in cellular metabolism, facilitating the transfer of electrons and hydrogen ions essential for energy production through redox (reduction-oxidation) reactions. The human body synthesizes NAD+ naturally. These functions are central to energy conversion from nutrients. NAD+ also supports DNA repair, regulation of gene expression, and immune system activity, as described in research publications.
The compound contributes to sirtuin activation, linking it to studies of aging and metabolic regulation. Interest in NAD+ among athletes and recovery specialists reflects these biological associations. Administration of an endogenous coenzyme via externally sourced injections introduces specific safety and efficacy variables—primarily with respect to dosing control, product purity, and immunological response—that remain undercharacterized in peer-reviewed human research. If food grade NAD+ is used for injectable forms this presents quality challenges. Only accredited 503 compounding facilities can make injectable forms and they need to use pharmaceutical grade material to ensure sterility and avoid contamination risk.
Is NAD+ Permitted in Sports?
NAD+ does not appear as a named substance on the World Anti-Doping Agency (WADA) Prohibited List as of 2026. WADA’s S0 category, however, restricts non-approved substances, including experimental drugs, research chemicals, and unapproved peptides. According to WADA’s published guidance, compounds designated "research use only" or "not for human consumption" fall under this restriction regardless of how they are marketed. WADA may determine the prohibited status of any unapproved drug or substance at its discretion.
NAD+ products sold as research chemicals fall into a regulatory grey area due to sourcing and manufacturing concerns. Even if NAD+ itself is not currently prohibited, product context and potential for contamination with other banned substances can lead to anti-doping violations. Injectable NAD+ from unverified sources may fail to meet product integrity standards, exposing users to inadvertent anti-doping rule violations. Sale of injectable or nasal spray forms outside of a medical environment that is overseen by a doctor presents legal risks.
WADA applies further restrictions to intravenous infusions, limiting administration to no more than 100 mL within a 12-hour period unless performed as part of hospital treatment or under formal medical supervision. Many protocols involving NAD+ exceed this infusion threshold, often reaching 500ml to 1,000ml, increasing compliance risks apart from the substance in question. So theoretically, even though NAD+ is not prohibited by name it could be prohibited in practice based on the WADA IV infusion limit. In other sport environments the IV infusion limits may not apply.
Military Regulations and Potential Restrictions
Military substance use policies apply strict liability, meaning service members are responsible for any positive drug test regardless of intent. As detailed in the military drug testing overview, ingestion of contaminated supplements can result in disciplinary consequences and administrative actions irrespective of knowledge or intent. This is the primary concern for military service members who are considering NAD+ as the WADA IV infusion limit does not apply in the military.
The U.S. Department of Defense maintains a list of prohibited dietary supplement ingredients under Operation Supplement Safety (OPSS). While NAD+ is not specifically named there, the listing encompasses unapproved drugs and research-only chemicals. Compounds like BPC-157 have been formally prohibited, and OPSS provides regular advisories and guidance to service members. NAD+ products sold as research chemicals present a grey area from this perspective as the substance is not prohibited but the form may be. NMN, the precursor to NAD+, has run into regulatory issues as it was determined that it is not a legal dietary supplement ingredient as it was previously explored as a drug, which makes it illegal to make as a supplement according to DSHEA.
Department of Defense protocol mandates dietary supplement education for all personnel and restricts on-base retail sales of products that include banned agents. These requirements reduce access to noncompliant products even in cases where a compound is not expressly named on a banned list. OPSS points to third-party certification as a vital element to review when considering dietary supplements.
U.S. Special Operations Command has conducted internal research regarding NAD+ enhancers, including preclinical studies focused on safety and dosing as described in published reports. However, research activities under controlled protocols do not confer general approval or authorization for routine service member use. Existence of military-funded research does not constitute product approval for operational use.
Quality and Safety Considerations
The highest safety risk associated with NAD+ products currently involves product sourcing. Many NAD+ products intended for consumer or athlete use are sold as research chemicals, not as legal dietary supplements or drugs produced by FDA-registered pharmacies or pharmaceutical-grade manufacturers.
Products labeled as research chemicals typically indicate "not for human consumption" or "research use only." These disclaimers aim to avoid dietary supplement or medical product regulations but also mean that the products are not held to Good Manufacturing Practices (GMP), systematic sterility controls, or standardized contamination screening. The peptide marketplace analysis highlights absence of validated identity and contaminant testing within the research chemical and unapproved peptide sectors.
A CNN investigation documented ambiguities about whether research chemicals or unapproved peptides are sourced from regulated pharmacies in the United States or from foreign chemical manufacturers that do not operate under FDA quality assurance. Identified issues in the research chemical realm have included substandard purity, bacterial contamination, heavy metals, and the presence of non-active or substituted substances. Similar quality control concerns may also be relevant for non-peptide products sold as research chemicals, like NAD+.
The FDA has cited compounded NAD+ preparations for contamination, with impurity levels exceeding 20% in certain samples. NAD+ is susceptible to degradation under routine exposure to light and moisture, which necessitates strict handling and storage protocols for product safety and efficacy.
Prescription NAD+ injections available through 503 compounding pharmacies should be subject to FDA registration and Current Good Manufacturing Practices, and distribution occurs through authorized medical channels. These products are distinct from those marketed directly as research chemicals for consumer purchase.
Third-Party Testing Insights
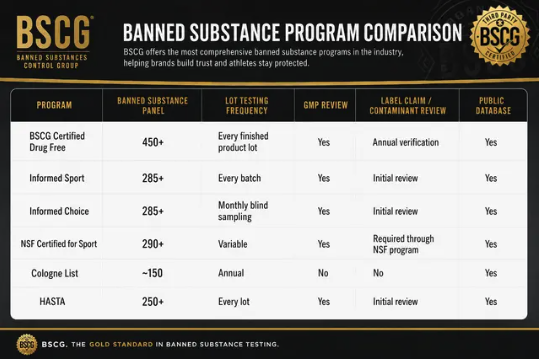
Independent supplement certification programs test for banned substances, verify label accuracy, and assess contaminant levels. As explained in the independent testing overview, these programs deploy ISO/IEC 17025-accredited laboratory protocols on dietary supplements prior to public distribution.
The BSCG Certified Drug Free program conducts comprehensive screens for more than 400 WADA prohibited drugs in addition to 50+ prescription and over-the-counter drugs, using low parts-per-billion detection limits on every lot of finished product. Lot-specific testing best addresses supply chain related adulteration or contamination and can drastically reduce the risk of inadvertent positive drug tests that may result from contaminated products. Limitations remain in third-party testing of research chemicals as such products are generally not accepted for certification by reputable third-party certification providers due to legal concerns.
For legal dietary supplement forms of NAD+ or precursors, independent testing can measure compound presence, concentration, and common contaminants, as well as identify banned substances from cross-contamination. However, current certification standards do not guarantee absolute exclusion of all banned substances. Certification reduces but does not eliminate inadvertent doping risks.
Major athletic, anti-doping, and military organizations, including the NFL Players Association, UFC, International Testing Agency, and DoD’s OPSS, recognize third-party certification programs such as BSCG Certified Drug Free, Informed Sport, and NSF Certified for Sport as tools to mitigate inadvertent doping risks. These programs pertain to dietary supplements that conform to regulatory frameworks, not to research chemicals outside these structures.
Implications for Brands, Compliance, and Manufacturing
Supplement brands that include NAD+ or similar compounds must account for regulatory risk and reputational considerations. The Dietary Supplement Ingredient Advisory List clarifies that many investigational, drug-like ingredients do not qualify as permitted actives for supplement certification. BSCG does not permit certification of supplements containing ingredients on this advisory list.
Brands considering NAD+ inclusion need robust quality assurance systems, including third-party GMP facility auditing, systematic testing for product identity and contaminants, and lot-specific assessment for banned substances. These processes are evidence of due diligence in protecting brand and consumer interests that are often missing in product forms sold as research chemicals or injectables. Third-party certification provides a way for legal supplement brands to showcase the quality of their products to consumers.
Brands that are looking to sell NAD+ must understand the legal and quality control concerns and account for them if they are going to sell NAD+ products legally. Nasal sprays, injectables, and IV infusion forms are not allowed to make the same structure function claims as dietary supplements, but they often do. This presents claims related FDA and FTC compliance risks.
An Evolving Story in Sport and Military Settings
Regulation of NAD+ and other research chemicals remains dynamic. WADA’s annual Prohibited List increasingly addresses research chemicals through growing categories such as metabolic modulators and non-approved substances, as detailed in a comprehensive WADA review.
Military regulatory bodies are revising policy frameworks. Department of Defense directives now specify supplement-use education requirements, retailer restrictions for prohibited substances, and guidance designed to reduce supplement-associated risks among service members. Third-party certification programs are recommended as a way to pro-actively protect military service members or athletes.
Research into NAD+ continues in academic and clinical settings, such as investigations of its roles in neurodegeneration, immune modulation, and human aging. Translational gaps persist between rigorously controlled research and unregulated product sales.
Athletes, military personnel, and supplement brands should treat any substance that exists within regulatory uncertainty, that is sold for research or administered off-label by injection, or is not explictly cleared by regulatory authorities, as inherently high risk until further policy and scientific consensus develops. IV infusion daily limits are important for athletes to consider to ensure they don't violate WADA regulations as NAD+ IV infusions often utilize 500ml or 1,000ml.
Frequently Asked Questions
Is NAD+ banned by WADA or sport organizations?
NAD+ does not appear by name on the WADA Prohibited List as of 2026. WADA’s S0 category covers non-approved substances, including research chemicals, potentially encompassing NAD+ products originating outside pharmaceutical channels. WADA has a 100ml daily IV infusion limit that may be violated by the typical IV infusion protocols for NAD+ that may range from 500ml to 1000ml per session.
Can military service members use NAD+ products?
NAD+ is not specifically banned in the military, but research chemical forms create a grey area as research chemicals are a concern in the military. Strict liability applies in military drug testing. Service members may face disciplinary action arising from consumption of contaminated or mischaracterized products that can lead to inadvertent positive drug tests making third-party certification an essential consideration.
What are the main quality concerns with NAD+ products?
NAD+ products from research chemical suppliers are not subject to Good Manufacturing Practices, sterility evaluation, or comprehensive contaminant screening. Typical risks include reduced purity, microbial contamination, heavy metal presence, and loss of efficacy due to inadequate storage conditions.
Can third-party testing detect NAD+ or verify its safety?
Third-party labs can confirm NAD+ identity and dosage, test for common contaminants, and screen for presence of banned substances. Third-party certification of legal NAD+ supplements can address such quality control concerns by verifying product integrity and quality and testing for hidden banned substances.
Should supplement brands include NAD+ in their products?
It is legal to sell NAD+ as a dietary supplement, but perhaps not as a research chemical. While oral availability reduces bioavailability it is the only legal way to sell NAD+ if you want to make structure function claims related to anti-aging or recovery. While injectable or nasal spray forms may seem attractive they come with increased risk of legal and regulatory scrutiny especially if they make structure function claims that are not allowed with the alternative forms.